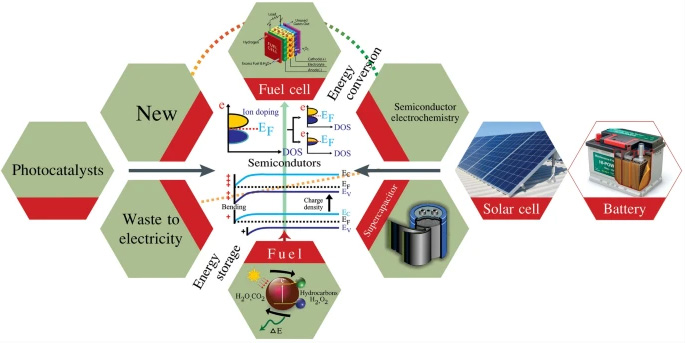
Semiconductors and the associated methodologies applied to electrochemistry have recently grown as an emerging field in energy materials and technologies. For example, semiconductor membranes and heterostructure fuel cells are new technological trend, which differ from the traditional fuel cell electrochemistry principle employing three basic functional components:anode, electrolyte, and cathode. The electrolyte is key to the device performance by providing an ionic charge flow pathway between the anode and cathode while preventing electron passage. In contrast, semiconductors and derived heterostructures with electron (hole) conducting materials have demonstrated to be much better ionic conductors than the conventional ionic electrolytes. The energy band structure and alignment, band bending and built-in electric field are all important elements in this context to realize the necessary fuel cell functionalities. This review further extends to semiconductor-based electrochemical energy conversion and storage, describing their fundamentals and working principles, with the intention of advancing the understanding of the roles of semiconductors and energy bands in electrochemical devices for energy conversion and storage, as well as applications to meet emerging demands widely involved in energy applications, such as photocatalysis/water splitting devices, batteries and solar cells. This review provides new ideas and new solutions to problems beyond the conventional electrochemistry and presents new interdisciplinary approaches to develop clean energy conversion and storage technologies.
Bin Zhu, Liangdong Fan, Naveed Mushtaq, Rizwan Raza, Muhammad Sajid, Yan Wu, Wenfeng Lin, Jung-Sik Kim, Peter D. Lund, Sining Yun
. Semiconductor Electrochemistry for Clean Energy Conversion and Storage[J]. Electrochemical Energy Reviews, 2021
, 4(4)
: 757
-792
.
DOI: 10.1007/s41918-021-00112-8


